Prode Properties
Properties of pure fluids and mixtures, multi phase equilibria, process simulation, software
Title : phase envelope, diagram, curve, curves, natural gas dew point, hydrate formation, Excel Matlab Mathcad software
Contact Prode
Prode Properties is the thermodynamic library created by Prode, it has been verified by Prode and other companies for more than 25 years of service, there are version for Windows, Linux, Android and client / server installations
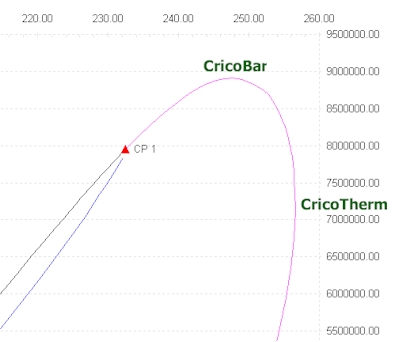
Application example : vapor-liquid-liquid phase envelope
Prode Properties can calculate different types of phase diagrams
- vapor-liquid phase envelope / phase diagram.
- vapor-liquid-liquid phase envelope / phase diagram.
- vapor-liquid-solid phase envelope / phase diagram.
Definitions
- Dew Point, given a equilibrium pressure the Dew Point Temperature is the maximum temperature in VLE area
- Bubble Point, given a equilibrium pressure the Bubble Point Temperature is the minimum temperature in VLE area
- CricondenBar, CricondenBar Pressure is the maximum pressure in VLE area
- Cricondentherm, Cricondentherm temperature is the maximum temperature in VLE area
Server Side (Linux server) online application examples
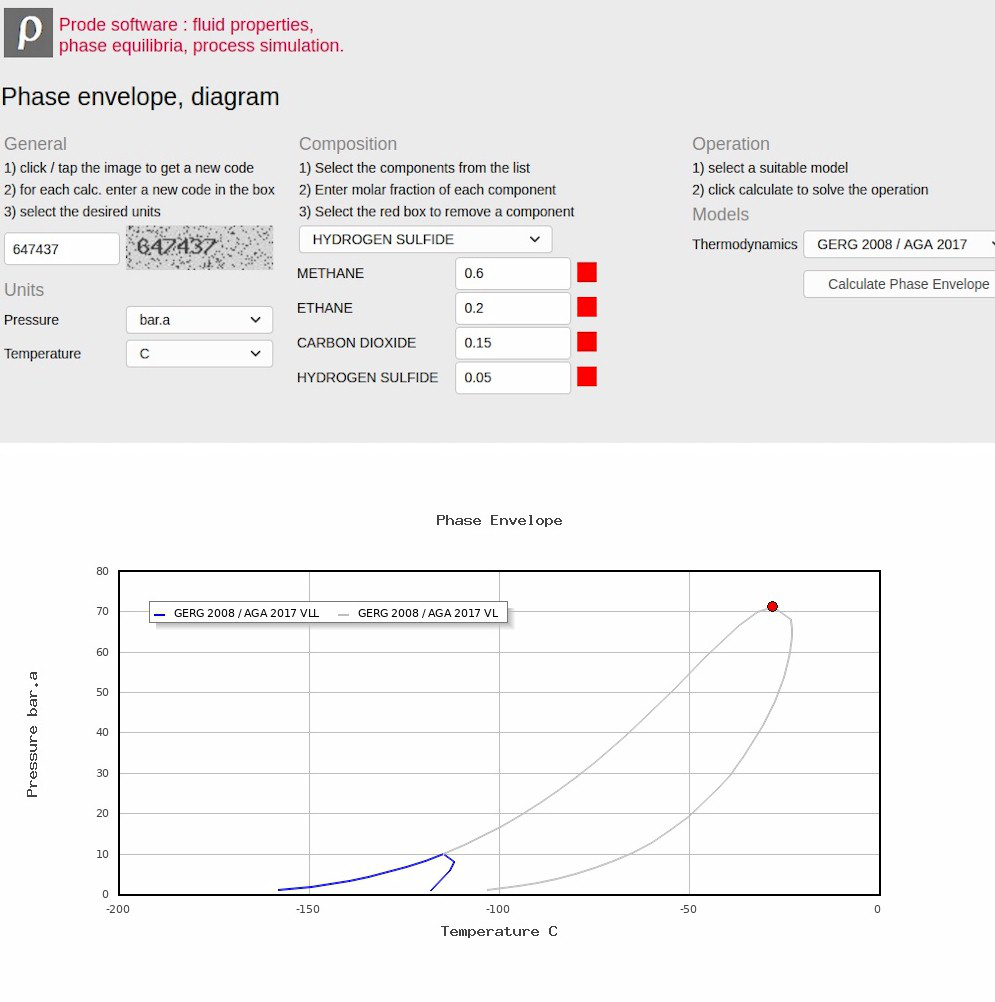
Local (Windows desktop) application example : create a phase diagram with Excel / Matlab / Mathcad
First step: define the stream (components, compositions etc.)
Properties includes a Stream editor which permits to access all informations (as compositions, operating conditions, models, options) for all streams which you need to define, to access the Stream editor from Excel Properties menu select Edit Properties
The Stream editor includes several pages, from the first page you can select a stream (Properties can store all the streams required to define a medium size plant) solve a series of flash operations and see the resulting compositions in the different phases, in this page select the stream you wish to define, for example the first.
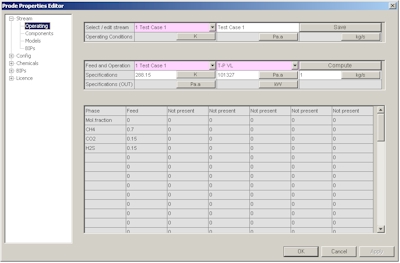
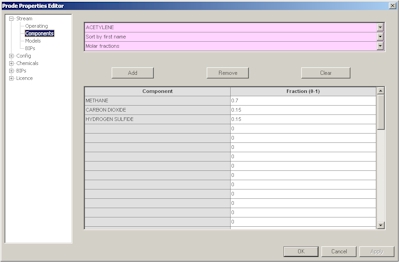
In the second page you can define a new composition or modify an existing composition, in this example we define C1 0.7 CO2 0.15 H2S 0.15 as molar fractions
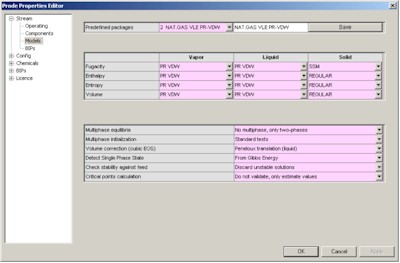
In the third page you can define the package (thermodynamic models and related options) , here we define API Soave Redlick Kwong.
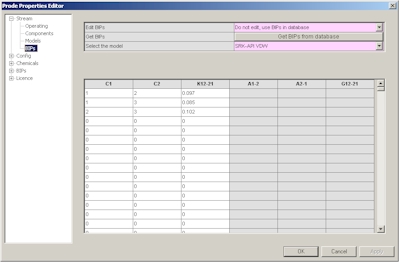
The fourth page provides access to BIP (Binary Interaction Parameters) for the different models, you can enter specific values or click on "Load BIPs" button to get the predefined BIPs from databank.
Finally we must save the new data, in the first page click on "Save" button, note that you can redefine the name of the stream as you wish (editing the cell near the button "Save"), you can define / modify many streams following the procedure described.
Once defined the stream you may wish to define the units which we wish to utilize in our problem, in stream editor go then to the "Units" dialog
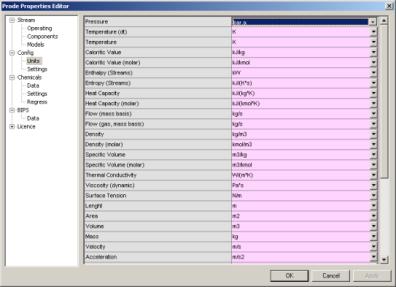
here you can select the units which you need for a specific problem, in this example for the pressure (first row) select Bar.a , notice that unit for temperature is K (but you can set the units which you prefer) then click on Ok button to accept new values and leave the Properties editor.
Now you are ready to use Properties for calculating all the properties which you need, however there is still a last thing to do if you do not wish to lose all data when leaving a Excel page, precisely to save data to a file, to save data to a file from Excel Properties menu select "Save a Archive"

then select the file "def.ppp" if you wish that Properties utilizes this data as default (this is the normal , recommended option), differently set a different name (you can for example define different names for different projects) but you will need to load that specific Archive before to make calc's for that project and since Excel reloads Properties with any new page this may result tedious...
Properties saves on the file also the units of measurement so you can define different streams and different units in different projects.
Now you can calculate all the properties which you need with the units which you prefer for all the streams defined in that project.
Second step: generate a vapor-liquid-liquid phase envelope in Excel.
For generating the phase envelope we?ll use a predefined Excel page distributed with Prode Properties, from Excel menu File->open , in Excel folder (Prode Properties installation) select the file phasenv.xls
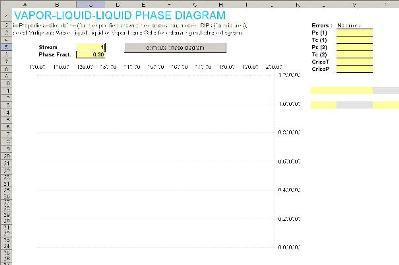
This page contains a little VBA code to tranfer the calculated equilibrium values (for the different vapor-liquid, liquid-liquid-vapor, vapor-liquid-liquid lines) from Prode Properties to Excel, if required you can easily modify the code for printing a series of lines with specified liquid or vapor fraction. To print a phase envelope you must define the stream (we select the first stream, which we defined with composition C1 0.7 CO2 0.15 H2S 0.15 , SRK as model and multiphase vapor+liquid option, the multiphase option instructs the procedure to generate a vapor-liquid-liquid phase envelope) we specify 0.3 value for liquid fraction, finally click on the button "calculate phase diagram".
Properties does all the work and the calculated equilibrium points including critical points, cricondentherm and cricondenbar are printed in Excel page for your analysis.
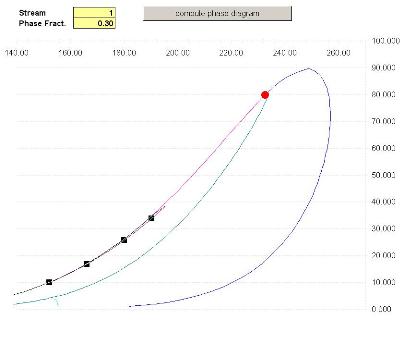
The phase envelope for this mixture shows a three phase area (notice the second line near the liquid-liquid-vapor bubble line and the third liquid-vapor-liquid line
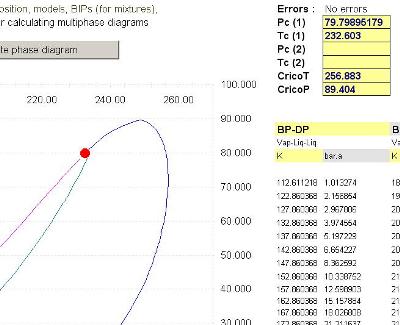
The calculated values for Critical Points, CricondenBar and CricondenTherm are available in Excel.
Note that Prode Properties calculates the TRUE critical points (not the estimated values), Prode Properties includes a proprietary procedure based on Gibbs minimization method.
the procedure allows to specify any value (from 0 -dew line- to 1 -bubble line-) for phase fractions, herebelow the example of a line with 0.05 liquid fraction.
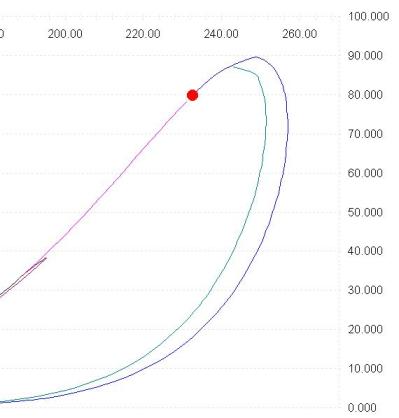
The Vapor-Liquid-Liquid phase envelope constitutes the ideal tool to investigate the phase equilibria for mixtures of hydrocarbons + water
The example shows the phase envelope for a 12 components (hydrocarbons + water) mixture, notice the large three phase area where liquid water is present.
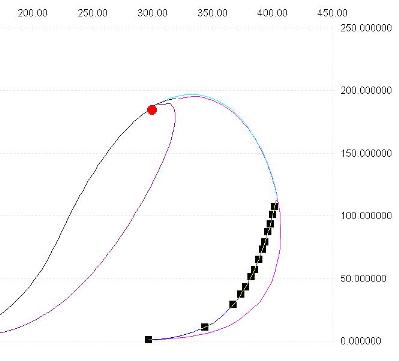
Note that even simple compositions can show a complex behaviour, herebelow is the example of the mixture with composition Methane 0.9 H2S 0.1 model API Soave Redlick Kwong.
The bubble line is a three phase (liquid-liquid-vapor) line, a vapor-liquid line connects the end of the liquid-liquid-vapor line with critical point, the dew line doesn't stop at critical point but continues, the blue line shows a liquid fraction of 0.05
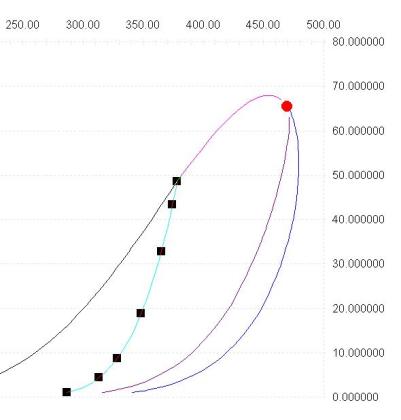
Note that Prode Properties includes methods for calculating critical points, CricondenBar and CricondenTherm in Excel cells, see the paragraph "Methods for thermodynamic calc?s" in operating manual for the details.
- methods StrPc() and StrTc() returns the critical pressure (or temperature) of the nth (from 1 to 5) critical point found.
- methods StrCBp() and StrCBt() returns the pressure (or temperature) of the CricondenBar (the equilibrium point with maximum pressure).
- methods StrCTp() and StrCTt() returns the pressure (or temperature) of the CricondenTherm (the equilibrium point with maximum temperature).
To get the value of critical pressure enter the macro =StrPc(1,1) where (1,1) refers to the stream 1 and first critical point detected, we enter this macro in B1, in B2 we enter the macro =StrTc(1,1) to calculate the critical temperature in the same way, in cells B3 and B4 we enter the macros = StrCBp(1) for CricodenBar pressure and = StrCTt(1) for CricodenTherm temperature.
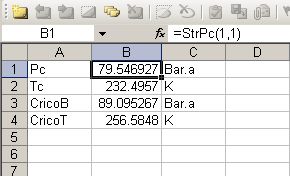
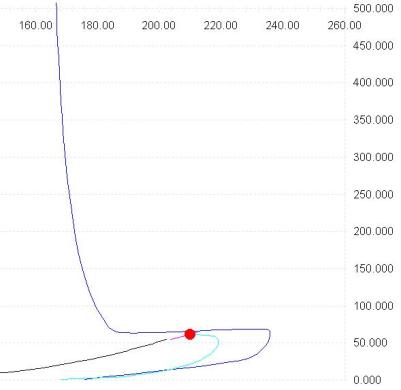
The phase envelope for this mixture (Methane 0.999 n-Butane 0.001) shows up to four saturation point pressures at the same temperature
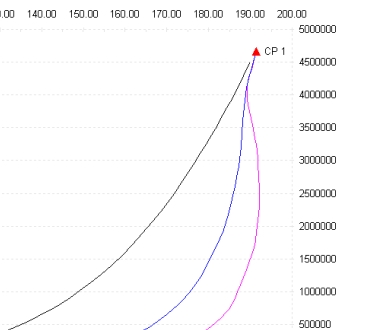
Observe the dew line, the red line below the critical point, there are up to three different equilibrium points at the same temperature (the area around 190 K), if you add the saturation point on the bubble line (black line) we have a total of four saturation point pressures at a given temperature, to calculate the points on the dew line we use the method:
double p = PfTF(integer stream, double t, double pf, int state, int n)
In cell B1 we define a value for the equilibrium temperature (190.208 K) , then in cells B2, B3, B4 we enter the macros
=PfTF(2,B1,0,1,1)
=PfTF(2,B1,0,1,2)
=PfTF(2,B1,0,1,3)
where the first value (2) is the stream which we defined, the second (cell B1) represents the temperature, the third (0) is the phase fraction (with 0 we specify 0% liquid or a point on dew line, the same would be by setting the state as gas and phase fraction as 1.0) the fourth (1) is the state (in Properties 0 = gas, 1 = liquid, 2 = solid) and the last is the required position (we require the points 1-3 along the dew line)
For additional infrmation about how to calculate equilibrium points go to the page Dew Points Natural Gas Hydrocarbons
Print a phase envelope in Matlab.
For calculating and printing the phase envelope we?ll use a predefined script distributed with Prode Properties, in Matlab command line type
>>phaseenvelope(1)
where (1) is the number of stream.
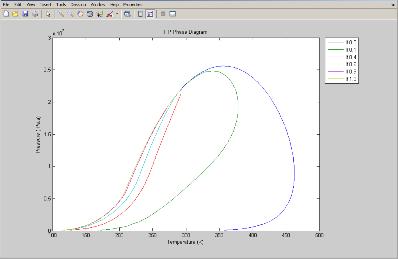
Prode Properties will calculate the phase envelope and print the result on Matlab, if you wish to obtain additional properties as cricondentherm, cricondenbar or critical points just enter the name of Properties method in Matlab, for example
>>StrPc(1)
will return the true critical pressure of stream 1.
Prode Properties, technical features overview (Windows version)
- Entirely written in C++ (since first edition, 1993)
- Up to 100 different streams with up to 50 components per stream (user can redefine)
- Several compilations of chemical data and BIPs are available, the user can add new components and BIPs
- Proprietary compilation with data for more than 1600 chemicals and 30000 BIPs
- flexible database format (support for up to 30 different correlations) works with all majour standards including DIPPR.
- Comprehensive set of thermodynamic models, base version includes
- Regular
- Wilson
- NRTL
- UNIQUAC
- UNIFAC
- Soave-Redlich-Kwong (standard and extended version with parameters calculated for best fitting of vapor pressure, density and enthalpy)
- Peng-Robinson (standard and extended version with parameters calculated for best fitting of vapor pressure, density and enthalpy)
- Benedict Webb Rubin (Starling) BWRS
- Steam Tables IAPWS 95
- ISO 18453 (GERG 2004)
- ISO 20765 (AGA 8 model)
- Lee-Kesler (Plocker) LKP
- CPA Cubic Plus Association (SRK and PR variants)
- Hydrates (Cubic Plus Association, Van Der Waals-Platteeuw)
- additional models as Pitzer, NRTL for electrolyte solutions, PC SAFT (with association), GERG (2008) etc.
- van der Waals and complex mixing rules to combine equations of state with activity models
- Huron Vidal
- Wong Sandler ( WS )
- Michelsen ( MHV2 )
- Tassios et al. ( LCVM )
- Base and Extended EOS versions with parameters calculated to fit experimental data from DIPPR and DDB
- Selectable units of measurement
- Procedure for solving fluid flow including multi phase equilibria and heat transfer
- Procedure for solving staged columns
- Rigorous solution of distillation columns, fractionations, absorbers, strippers...
- Procedure for calculating hydrate formation temperature and hydrate formation pressure
- hydrate phase equilibria based on different Van Der Waals-Platteeuw models
- Procedure for solving polytropic compression with phase equilibria
- Huntington method for gas phase
- Proprietary method for solving a polytropic process with phase equilibria
- Procedure for solving isentropic nozzle (safety, relief valve with single and two phase flow)
- HEM, Homogeneous Equilibrium
- HNE-DS, Homogeneous Non-equilibrium
- NHNE, Non-homogeneous Non-equilibrium
- Procedure for simulating fluid flow in piping (pipelines) with heat transfer
- Beggs and Brill and proprietary methods for single phase and multiphase fluid flow with heat transfer
- Procedure for fitting BIP to measured VLE / LLE / SLE data points (data regression)
- Procedure for fitting BIP to VLE values calculated with UNIFAC
- Functions for simulating operating blocks (mixer, gas separator, liquid separator) **
- Functions for accessing component data in database (the user can define mixing rules)
- gas / vapor-liquid-solid fugacity plus derivatives vs. temperature pressure composition
- gas / vapor-liquid-solid enthalpy plus derivatives vs. temperature pressure composition
- gas / vapor-liquid-solid entropy plus derivatives vs. temperature pressure composition
- gas / vapor-liquid-solid molar volume plus derivatives vs. temperature pressure composition
- Flash at Bubble and Dew point specifications and P (or T)
- Flash at given temperature (T) and pressure (P) multiphase vapor-liquid-solid, isothermal flash
- Flash at given phase fraction and P (or T), solves up to 5 different points
- Flash at given enthalpy (H) and P multiphase vapor-liquid-solid, includes adiabatic flash
- Flash at given enthalpy (H) and T multiphase vapor-liquid-solid, includes adiabatic flash
- Flash at given entropy (S) and P multiphase vapor-liquid-solid, includes isentropic flash
- Flash at given entropy (S) and T multiphase vapor-liquid-solid, includes isentropic flash
- Flash at given volume (V) and P multiphase vapor-liquid-solid, includes isochoric flash
- Flash at given volume (V) and T multiphase vapor-liquid-solid, includes isochoric flash
- Flash at given volume (V) and enthalpy (H) multiphase vapor-liquid-solid
- Flash at given volume (V) and entropy (S) multiphase vapor-liquid-solid
- Flash at given enthalpy (H) and entropy (S) multiphase vapor-liquid-solid
- Rigorous (True) critical point plus Cricondentherm and Cricondenbar
- Complete set of properties for different states
- gas density
- vapor density
- liquid density
- solid density
- gas Isobaric specific heat (Cp)
- vapor Isobaric specific heat (Cp)
- liquid Isobaric specific heat (Cp)
- gas Isochoric specific heat (Cv)
- vapor Isochoric specific heat (Cv)
- liquid Isochoric specific heat (Cv)
- gas cp/cv
- liquid cp/cv
- Gas heating value
- Gas Wobbe index
- Gas Specific gravity
- gas Joule Thomson coefficients
- vapor Joule Thomson coefficients
- liquid Joule Thomson coefficients
- gas Isothermal compressibility
- vapor Isothermal compressibility
- liquid Isothermal compressibility
- gas Volumetric expansivity
- vapor Volumetric expansivity
- liquid Volumetric expansivity
- gas Speed of sound
- vapor Speed of sound
- liquid Speed of sound
- vapor + liquid (HEM) Speed of sound
- gas Viscosity
- vapor Viscosity
- liquid Viscosity
- gas Thermal conductivity
- vapor Thermal conductivity
- liquid Thermal conductivity
- gas compressibility factor
- vapor compressibility factor
- liquid Surface tension
Typical applications
- Fluid properties in Excel, Matlab, Mathcad and other Windows and UNIX (**) applications
- Thermodynamics, physical, thermophysical properties
- Process simulation
- Heat / Material Balance
- Process Control
- Process Optimization
- Equipments Design
- Separations
- Instruments Design
- Realtime applications
- petroleum refining, natural gas, hydrocarbon, chemical, petrochemical, pharmaceutical, air conditioning, energy, mechanical industry