Prode Properties
multi phase vapor liquid solid hydrate formation software
Title : Hydrate formation, dissociation, inhibition, software, multi phase equilibria in Windows, Linux, Android...
Contact Prode
Hydrates in natural gas industry
Interest for hydrates began when researchers found that natural gas hydrates can block gas transmission lines even at temperatures above the ice point, after the discovery many researchers starting from Hammerschmidt, Deaton, Frost investigated the effects of inhibitors such as salts (chloride salts...) liquids (methanol, ethanol, glycols as mono ethylene glycol MEG etc.) on hydrates, thermodynamic inhibitors lower the freezing point and thereby reduce risk of hydrate formation.
Mechanisms of gas hydrate formation and inhibition
The necessary condition for hydrate formation is the presence of water (or ice), gas hydrates can form from dissolved gas or free gas. Thermodynamic inhibitors such as methanol, ethanol, mono ethylene glycol (MEG) etc. can reduce kinectics of nucleation, other inhibitors can reduce agglomeration of hydrate crystals.
Predicting gas hydrate formation
Several methods are available to predict hydrate formation conditions, the old methods (Wilcox, Katz etc.) allow direct solutions while more recent methods based on Van der Waals and Platteeuw theory require a computer software.
Solving multi phase equilibria including hydrate phase
The multi phase flash procedure in Prode Properties solves multi phase equilibria including solid and hydrate phases, given operating conditions and compositions the procedure calculates fugacities plus derivatives of each phase and the second order solver identifies the correct solution. A phase stability step removes unstable phases. The procedure doesn't require the presence of a free water phase and can include single or mixed salts (with electrolyte models).
Accuracy
Prode Properties includes two different models for hydrate phase, a simplified method and a complex method, both based on Van der Waals and Platteeuw theory. Prode Properties allows to select differnt models for vapor, liquid and solid fugacities.
Here, errors (for about 100 points) are determined as difference in hydrate formation pressure calculated by Prode Properties with CPA-PR model vs. measured data sets.
You can contact Prode to receive the complete data set with calculated data and the reference in literature.
- Simplified method, SI, SII, SH structures, mixtures with 2 or more formers, errors on predicted hydrate formation pressure :
- SI max error < 10% average error 4%
- SII max error < 20% average error 6%
- SH max error < 10% average error 4%
- Complex method, SI, SII, SH structures, mixtures with 2 or more formers, errors on predicted hydrate formation pressure :
- SI max error < 7% average error 3%
- SII max error < 12% average error 4%
- SH max error < 6% average error 3%
Prode Properties is the thermodynamic library created by Prode, it has been verified by Prode and many other companies for more than 25 years of service, there are version for Windows, Linux, Android and client / server installations
Server Side (Linux server) online application examples
- Phase envelopes / diagrams
- Compare phase envelopes / diagrams with different models
- Hydrate formation
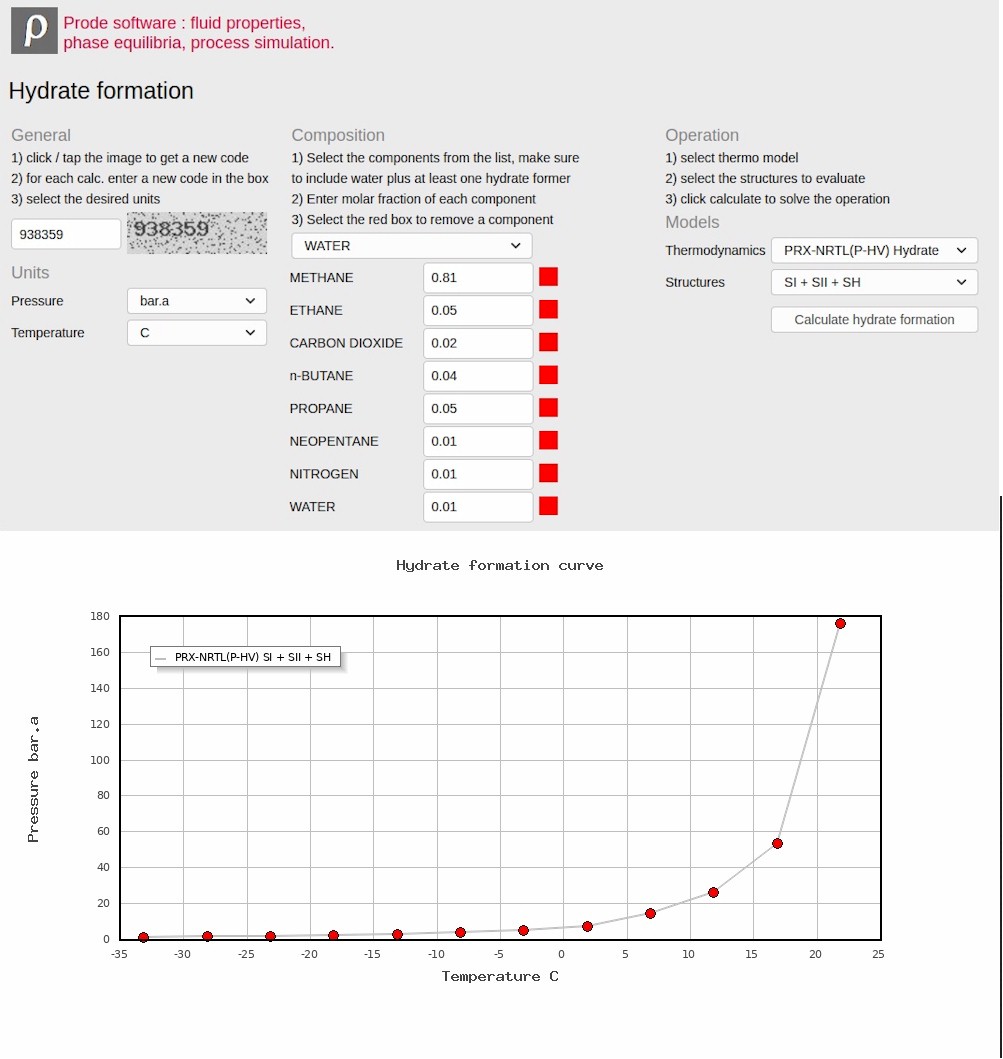
Local (desktop) application example : multi phase equilibria including hydrates
Calculate multiphase equilibria with solids (hydrate formation) in Prode Properties Editor (available in all versions of Prode Properties), the Editor allows to solve many unit operations including Vapor-Liquid-Solid (with Hydrate phase) flash, the selected model (PRXCPA-NRTL(P-HV)) at 277 K and 20 Bar.a predicts a hydrate phase
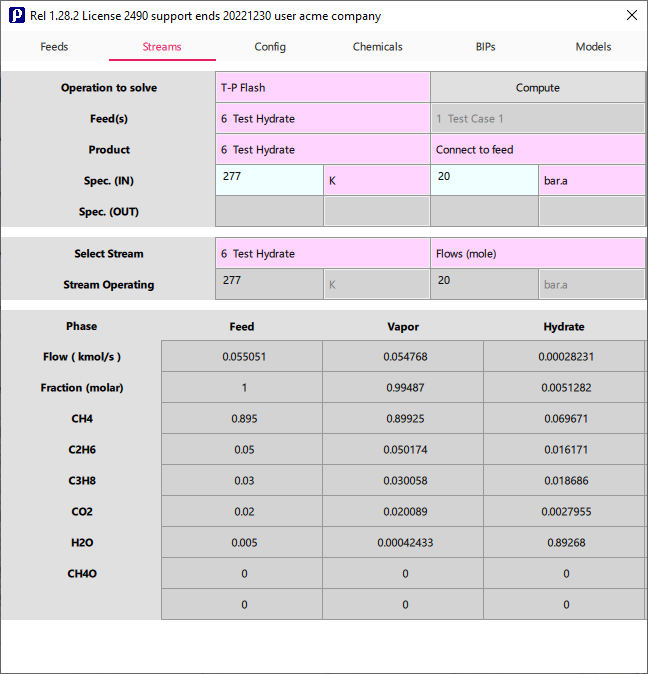
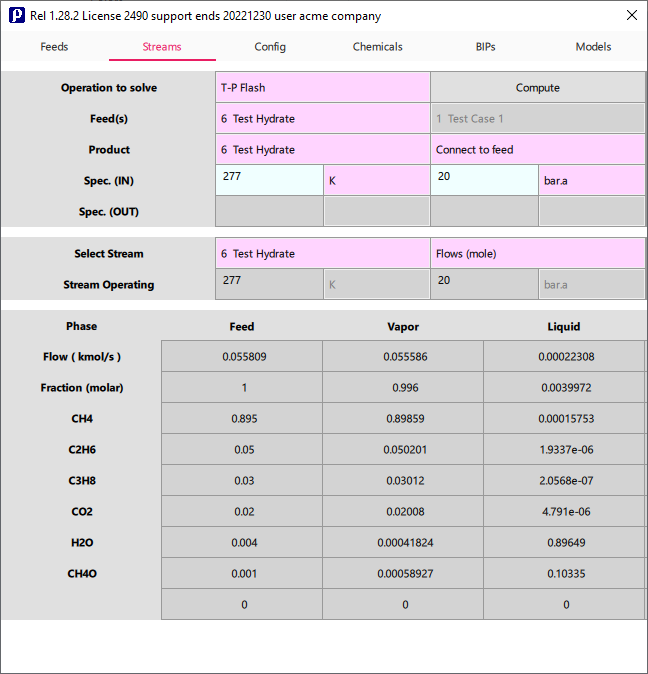
Evaluate how much methanol (or another inhibitor) is required to prevent hydrate formation at specified conditions
You can evaluate how the addition of a small amount of methanol (inhibitor) can increase the formation pressure for hydrate phase, we recalculate phase equilibria including a 0.001 mole fraction of methahol, as result we must increase the operating pressure to detect a hydrate phase.
To improve the accuracy of phase equilibria in presence of inhibitors it is recommended to adopt specific BIPs calculated from SLE data points (you may contact Prode for assistance)
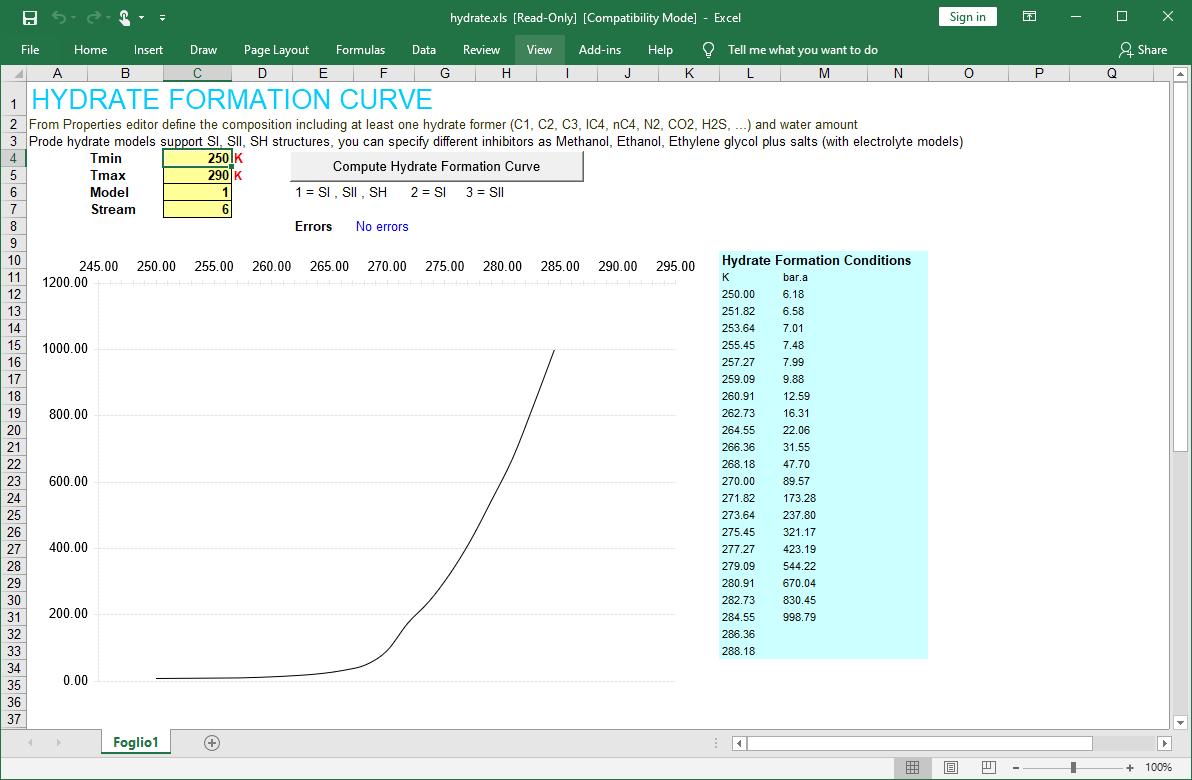
Hydrate formation temperature curve and hydrate formation pressure curve in Excel
With Prode Properties you can calculate the hydrate formation temperature curve or the hydrate formation pressure curve and print a graph in Excel.
- open the example page hydrate.xls included in distribution
- select the stream 6, edit the stream to modify the composition, if required
- click on Calculate Hydrate Formation Curve button to generate the graph
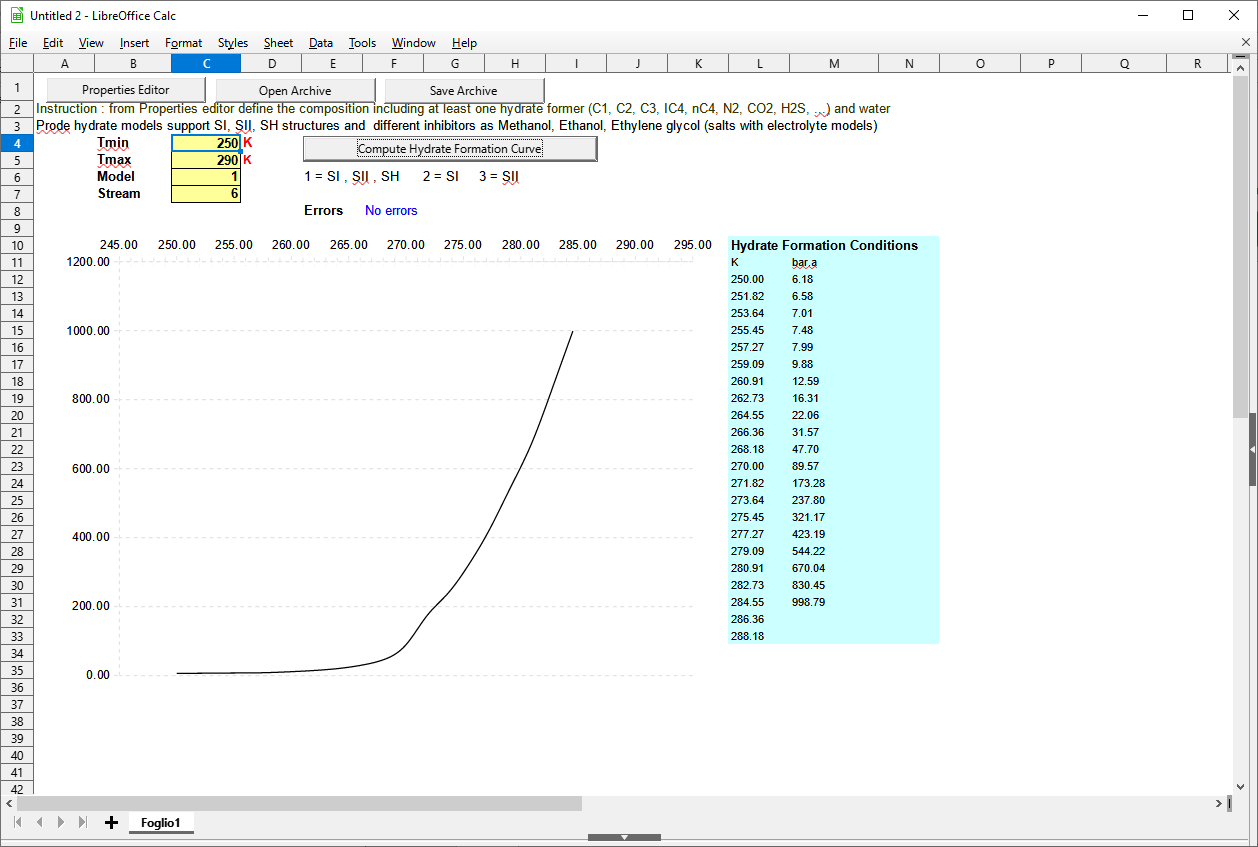
Hydrate formation temperature curve and hydrate formation pressure curve in OpenOffice (Libreoffice)
In a similar way you can calculate the hydrate formation temperature curve or the hydrate formation pressure curve and print a graph in Open Office (LibreOffice).
- open the example page hydrate.xls included in distribution
- select the stream 6, edit the stream to modify the composition, if required
- click on Calculate Hydrate Formation Curve button to generate the graph
Technical features overview
- Entirely written in C++ with native support for multiple threads, no limits on number of concurrent (parallel) threads
- Comprehensive set of thermodynamic models with specific models for hydrate phase equilibria :
- Standard cubic EOS as Peng Robinson, Soave Redlich Kwong etc.
- EOS based on corresponding states theory as Lee Kesler (Plocker) etc.
- Extended EOS with complex mixing rules (Huron-Vidal, Michelsen, Wong-Sandler etc.)
- Extended EOS with association contribute and complex mixing rules
- Van Der Waals-Platteeuw
- additional models as Pitzer, NRTL for electrolyte solutions etc.
- standards as GERG 2008 (AGA 2017), IAPWS 95 etc.
- Complete set of properties for the different states
- Thermodynamic and transport properties for vapor, liquid, solid (hydrate, asphaltenes etc.) phases
- Comprehensive set of operation units
- Flash operations with multiphase (vapor,liquid,solid) equilibria
- Distillation columns
- ...
Typical applications
- Fluid properties for pure fluids and mixtures
- Process simulation
- Heat / Material Balance
- Process Control
- Process Optimization
- Equipments Design
- Separations
- Instruments Design
- Realtime applications
- petroleum refining, natural gas, hydrocarbon, chemical, petrochemical, pharmaceutical, air conditioning, energy, mechanical industry